Search OneLab Network Past Events
2023

Diagnostic Excellence: A New Quality Tool to Prevent Blood Culture Contamination
Laboratory analysis of blood cultures is vital to the accurate and timely diagnosis of bloodstream infections. However, the reliability of your testing depends on clinical compliance with collection procedures that limit the risk of inconclusive or incorrect results.

Empowering Healthcare: Bridging Gaps, Building Futures with the Clinical Laboratory Development Program
The shortage of clinical diagnostic laboratory personnel directly impacts the quality of healthcare services, patient outcomes, and the healthcare system's ability to respond to emerging health challenges and technological advancements effectively.

CLIA Proficiency Testing (PT) Final Rule, CMS-3355-F
This presentation will provide information on the CLIA Proficiency Testing (PT) Final Rule which was published in the Federal Register Notice on July 11, 2022.
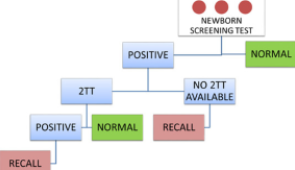
Oregon State Public Health Laboratory Shares Lessons Learned from Its Newborn Screening Program
The history of newborn screening (NBS) demonstrates the strength and integrity of its mission: to identify children with congenital conditions who need treatment to prevent adverse outcomes.

Biosafety Practices and Reporting Occupational Exposures for Select Agents and Toxins (Part 2)
This basic-level webinar is designed to help clinical and veterinary laboratory professionals understand how to report select agent and toxin identifications and recognize and report exposure incidents.

Recognizing, Identifying, and Reporting the Identification of Select Agents and Toxins
This beginner-level webinar is for clinical and veterinary laboratory professionals to understand how to report select agent and toxin identifications and recognize and report exposure incidents.
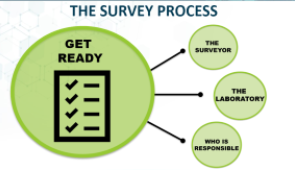
The Survey Process: What You Need to Know for Your CLIA Survey
Preparing for laboratory survey? Laboratory staff are invited to join this webinar, where Marranda Scott, a Clinical Laboratory Scientist with the CDC Division of Laboratory Systems, will review processes and requirements for a CLIA laboratory survey.
Pipeline and Hazardous Materials Safety Administration (PHMSA) Training Program Requirements: Shipping Hazmat such as Cat A
The U.S. Department of Transportation provides helpful information about creating and implementing a laboratory hazmat plan.

Navigating Laboratory Education and Training Needs: An Open Forum
Start your year by collaborating and sharing your training and education needs through CDC’s OneLab Network. Do you have laboratory guidance needs? Are there training materials you are interested in developing for your laboratory staff?
